When you access an area of the FDA that requires authorization, the system will require you to log in with your NYU netID and password. All users can register to become subscribers to public collections. Some restricted functions, such as content submission, require authorization from the community.
My FDA is a personal page that is maintained for each member. This page can contain a list of items that are in the submission process for a particular member, or a task list of items that need attention such as editing, reviewing, or checking.
Edit Profile allows you to change your password and change the information we have for you. You must be authenticated with your log-in to change any of your personal information.
| SEARCH | top |
To search all of the FDA, use the the Search option on the top right corner or the search box in the middle of the home page.
To limit your search to a specific community or collection, navigate to that community or collection and use the search bar on that page.
The FDA uses the Solr/Lucene search engine. Here are some search hints:
The word(s) you enter in the search box will be searched against each item’s descriptive metadata (including title, author, subject, series, etc.). Additionally, the word(s) you enter will also be searched across the full text of the item itself, if the item has been optimized for optical character recognition (OCR).
What is not searched - Stop Words
The search engine ignores certain words that occur frequently in English, but do not add value to the search. These are:"a", "and" , "are" , "as" , "at" , "be" , "but" , "by" , "for" , "if" , "in" , "into",
"is" ,"it" ,"no" , "not" , "of" , "on" , "or" , "such", "the" , "to" , "was"
Truncation
Use an asterisk (*) after a word stem to get all hits having words starting with that root, for example:For example: Select* will retrieve selects, selector, selectman, selecting.
Stemming
The search engine automatically expands words with common endings to include plurals, past tenses, etc.Phrase Searching
For example: "organizational change"
To search using multiple words as a phrase, put quotation marks (") around the phrase.Exact word match
Put a plus (+) sign before a word if it MUST appear in the search result. For instance, in the following search the word "training" is optional, but the word "dog" must be in the result.For example: +dog trainingEliminate items with unwanted words
Put a minus (-) sign before a word if it should not appear in the search results. Alternatively, you can use NOT. This can limit your search to eliminate unwanted hits.For example: In the searchtraining -cat or training NOT cat you will get items containing the word "training", except those that also contain the word "cat".Boolean searching
The following Boolean operators can be used to combine terms. Note that they must be CAPITALIZED!
AND - to limit searches to find items containing all words or phrases combined with this operator, e.g.
cats AND dogs will retrieve all items that contain BOTH the words "cats" and "dogs".OR - to enlarge searches to find items containing any of the words or phrases surrounding this operator
cats OR dogs will retrieve all items that contain EITHER the words "cats" or "dogs".Parentheses can be used in the search query to group search terms into sets, and operators can then be applied to the whole set. For example:
| ADVANCED SEARCH | top |
The advanced search page allows you to specify the fields you wish to search, and to combine these searches with the Boolean "and", "or" or "not".
You can restrict your search to a community by clicking on the arrow to the right of the top box. If you want your search to encompass all of the FDA, leave that box in the default position.
Enter the phrase you are searching for in the search box. You can further refine the search by specifying what the title, author, subject or date issued should equal, contain, not equal or not contain.
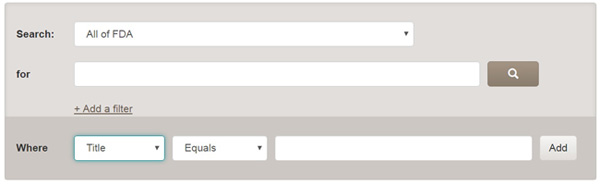
Note: You must use the input boxes in order. If you leave the first one blank your search will not work.
| COMMUNITIES | top |
Within each community there can be an unlimited number subcommunities and an unlimited number of collections. Subcommunities and collections correspond to administrative entities such as departments, labs, research centers, and individual researchers. FDA content lives at the collection level, and each collection may contain an unlimited number of items.
Each community has its own entry page displaying information, news and links reflecting the interests of that school/college, as well as a descriptive list of collections within the community.
For the FDA, the the hierarachy of structure is as follows -- NYU school or division --> Collection of faculty member/project --> Individual item.
| COLLECTIONS | top |
Communities can maintain an unlimited number of collections in the FDA. Collections can be organized around a topic, or by type of information (such as working papers or datasets) or by any other sorting method a community finds useful in organizing its digital items. Collections can have different policies and workflows.
Each FDA collection has its own entry page displaying information, news and links reflecting the interests of users of that collection.
| SUBMIT | top |
Stopping during the Submission Process:
At any point in the submission process you can stop and save your work for a later date by clicking on the "cancel/save" button at the bottom of the page. The data you have already entered will be stored until you come back to the submission, and you will be reminded on your "My FDA" page that you have a submission in process. If somehow you accidentally exit from the submit process, you can always resume from your "My FDA" page. You can also cancel your submission at any point.
Progress Bar - Buttons at Top of Page:
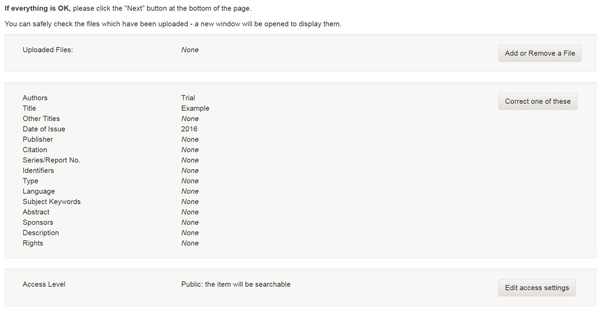
At the top of the submit pages you will find 6 rectangular buttons representing each step in the submission process. As you move through the process these buttons will change color. Once you have started you can also use these buttons to move back and forth within the submission process by clicking on them. You will not lose data by moving back and forth.

Select Collection:
Click on the arrow at the right of the drop-down box to see a list of Collections. Move your mouse to the collection into which you wish to add your item and click.
(If you are denied permission to submit to the collection you choose, please contact contact archive.help@nyu.edu for more information.)
You must be authorized by a community to submit items to a collection. If you would like to submit an item to the FDA, but don't see an appropriate community, please contact contact archive.help@nyu.edu to find out how you can get your community set up in FDA.
Click on the "Next" button to proceed, or "Cancel/Save" button to stop and save or cancel your submission.
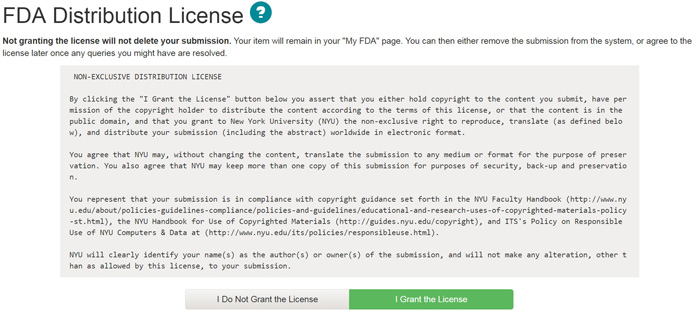
| STEP 1: License | top of submit | top |
The FDA requires agreement to this non-exclusive distribution license before your item can appear in the Archive. Please read the license carefully. If you have any questions, please contact archive.help@nyu.edu.
There are two ways of uploading the file you want. You can either select the file using the 'Open' option or you can drag and drop the file into the upload window.
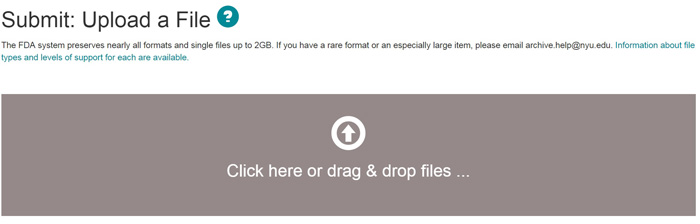
Click on the "next" button to proceed, or "cancel/save" button to stop and save or cancel your submission. You can also choose 'Skip File Upload' to add the file at a later time.
| STEP 3: Describe | top of submit | top |
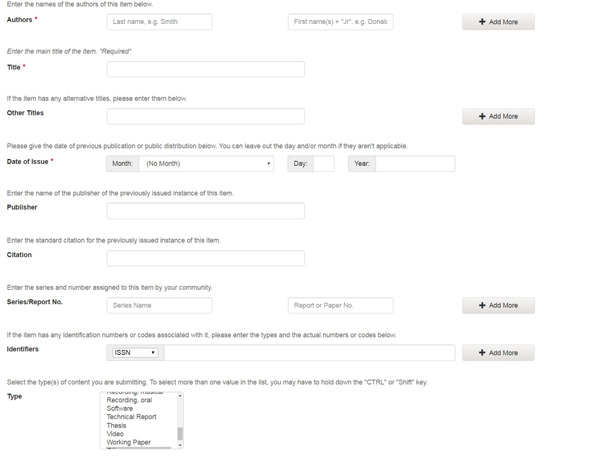
The information you fill in on this screen will form the metadata record that will enable users to retrieve your item using search engines. The richer the metadata, the more "findable" your item will be, so please take the time to fill in as many fields as are applicable to your item. The Author, Title and Date of Issue (year) fields are mandatory.
Author:
This can be a person, organization or service responsible for creating or contributing to the content of the item. By clicking on the "Add More" button you can add as many authors as needed.
If the author is an organization, use the last name input box for the organization name.
Title:
Enter the full and proper name by which this item should be known. All FDA items must have a title!
Other Titles:If your item has a valid alternative title, for instance, a title in another language or an abbreviation, then enter it here.
Date of Issue:
If your item was previously published or made public, enter the date of that event here. If you don't know the month, leave the default "no month"; otherwise select a month from the drop-down box. If you don't know the exact day, leave that box empty.
Publisher:
Enter the name of the publisher of this item.
Citation:
Enter citation information
for this item if it was a journal article or part of a larger work, such as
a book chapter. For journal articles, include the journal title, volume
number, date and paging.
For book chapters, include the book title, place of publication, publisher
name, date and paging.
Series/Report No.:
Some of the collections in the FDA are numbered series such as technical reports or working papers. If this collection falls into that category, then there should be a default value in the Series Name box which you should not change, but you will have to fill in the assigned number in the Report or Paper No. input box.
Identifiers:
If you know of a unique number or code that identifies this item in some system, please enter it here. Click on the arrow to the right of the input box, and select from one of the choices in the drop down menu. The choices refer to:
Govt.doc # - Government Document Number - e.g. NASA SP 8084
DOI - Digital Object Identifier - e.g. 10.10.1038/nphys1170
ISBN - International Standard Book Number - e.g. 0-1234-5678-9
ISSN - International Standard Serial Number - e.g. 1234-5678
ISMN - International Standard Music Number - e.g. M-53001-001-3
URI - Universal Resource Identifier - e.g.. http://www.dspace.org/help/submit.html
Other - An unique identifier assigned to the item using a system other than the above
Type:
Select the type of work (or genre) that best fits your item. To select more than one value in the list, you may have to hold down the "ctrl" or "shift" key.
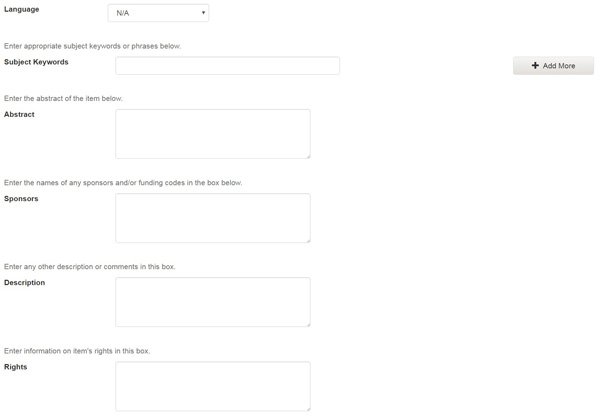
Language:
Select the language of the intellectual content of your item. If the default (English - United States) is not appropriate, click on the arrow on the right of the drop down box to see a list of languages commonly used for publications, e.g.
If your item is not a text document and language is not applicable as description, then select the N/A choice.
Subject Keywords:
Please enter as many subject keywords as are appropriate to describe this item, from the general to the specific. The more words you provide, the more likely it is that users will find this item in their searches. Use one input box for each subject word or phrase. You can get more input boxes by clicking on the "add more" button. Examples:
Your community may suggest the use of a specific vocabulary, taxonomy, or thesaurus. If this is the case, please select your subject words from that list.
Abstract:
You can either cut and paste an abstract into this box, or you can type in the abstract. There is no limit to the length of the abstract. We urge you to include an abstract for the convenience of end-users and to enhance search and retrieval capabilities.
Sponsors:
If your item is the product of sponsored research, you can provide information about the sponsor(s) here. This is a freeform field where you can enter any note you like.
Description:
Here you can enter any other information describing the item you are submitting or comments that may be of interest to users of the item.
Rights:
Here you can enter any information about the rights of the use of the item you are submitting.
Click on the "next" button to proceed, or "cancel/save" button to stop and save or cancel your submission.
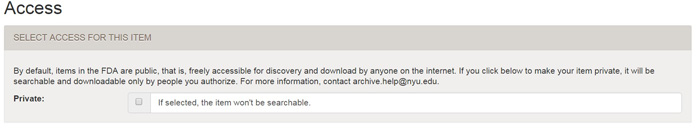
| STEP 4: Access | top of submit | top |
On the Access page, check the box only if you want your item to be private. By default, all items in the FDA are public (and we strongly encourage this because it makes your work accessible to wider audience).